
Chemistry, 18.01.2021 18:10 mallorytaylor279
2.In a titration, 25.00 cm3 of a solution of hydrochloric acid reacted with 18.40 cm3 of sodium hydroxide solution of concentration 0.15 mol/dm3.
The equation which represents the reaction is:
HCl + NaOH > NaCl + H2O
Calculate the concentration of hydrochloric acid in mol/dm3

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1


Chemistry, 22.06.2019 17:30
To find the enthalpy of a reaction in the lab, you measured the of the reactants and the change during the reaction.
Answers: 1
You know the right answer?
2.In a titration, 25.00 cm3 of a solution of hydrochloric acid reacted with 18.40 cm3 of sodium hydr...
Questions

History, 08.12.2021 07:40

Computers and Technology, 08.12.2021 07:40



Mathematics, 08.12.2021 07:40


Health, 08.12.2021 07:40

Mathematics, 08.12.2021 07:40

Mathematics, 08.12.2021 07:40

Mathematics, 08.12.2021 07:40



Physics, 08.12.2021 07:40


Mathematics, 08.12.2021 07:40




Mathematics, 08.12.2021 07:40

Mathematics, 08.12.2021 07:40

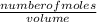 =
= 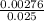 = 0.11mol/dm³
= 0.11mol/dm³

