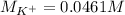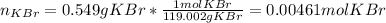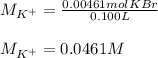Chemistry, 18.01.2021 14:00 makayyafreeman
Suppose of potassium bromide is dissolved in of a aqueous solution of silver nitrate. Calculate the final molarity of potassium cation in the solution. You can assume the volume of the solution doesn't change when the potassium bromide is dissolved in it. Round your answer to significant digits.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:30
The is a particle with one unit of positive charge a. proton b. positron c. electron d. nucleus awnser quick it is a important science test!
Answers: 2

Chemistry, 22.06.2019 09:00
Given the following reaction: c3h8+5o2=3co2+4h20 how many grams of co2 will be produced 7 g of c3h8 and 98 g of o2
Answers: 1


You know the right answer?
Suppose of potassium bromide is dissolved in of a aqueous solution of silver nitrate. Calculate the...
Questions


Mathematics, 24.12.2019 15:31



History, 24.12.2019 15:31

Biology, 24.12.2019 15:31

Biology, 24.12.2019 15:31

Biology, 24.12.2019 15:31

Mathematics, 24.12.2019 15:31

History, 24.12.2019 15:31


History, 24.12.2019 15:31

Mathematics, 24.12.2019 15:31

English, 24.12.2019 15:31

Mathematics, 24.12.2019 15:31