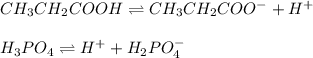Chemistry, 18.01.2021 14:00 zalyndevola
Propionic acid (CH3CH-COOH) has a K, 1.3 x 10^-5 and phosphoric acid (H3PO2) has a Ka = 7.5 x 10^-3
Choose the conjugate base for each.
A. CH3CH2COO2- for CH3CH2COOH; HPO4 2- for H3PO4
B. CH3CH2CO- for CH3CH2COOH; H2PO3 - for H3PO4
C. CH3CH2COOH2 for CH3CH2COOH; H4PO4 for H3PO4
D. CH3CH2COO- for CH3CH2COOH; H2PO4 - for H3PO4

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
When a spring is compressed, the energy changes from kinetic to potential. which best describes what is causing this change?
Answers: 3

Chemistry, 23.06.2019 01:00
How does carbon monoxide pose the greatest threat to humans? a. it can be produced by wood fires. b. it can be produced by home furnaces. c. it is produced by acid rain. d. it is produced by modern automobiles.
Answers: 2

Chemistry, 23.06.2019 02:30
When the ionic compound nabr dissolves in water, br– ions are pulled into solution by the attraction between what two particles? a. the na+ and br– ions b. the na+ ion and the negative end of a water molecule c. the br– ion and the positive end of a water molecule d. the br– ion and the negative end of a water molecule
Answers: 1

Chemistry, 23.06.2019 15:20
Which element below could be an isotope of berylliumsodium-10beryllium-10boron-9carbon-9
Answers: 2
You know the right answer?
Propionic acid (CH3CH-COOH) has a K, 1.3 x 10^-5 and phosphoric acid (H3PO2) has a Ka = 7.5 x 10^-3...
Questions

History, 05.07.2021 09:20

Biology, 05.07.2021 09:20


Mathematics, 05.07.2021 09:20

World Languages, 05.07.2021 09:20

Physics, 05.07.2021 09:20

Mathematics, 05.07.2021 09:20



Mathematics, 05.07.2021 09:20

Chemistry, 05.07.2021 09:20

Mathematics, 05.07.2021 09:20






Mathematics, 05.07.2021 09:20

Chemistry, 05.07.2021 09:20

Biology, 05.07.2021 09:20