
Hydrobromic acid dissolves solid iron according to the following reaction:
Fe(s)+2HBr(aq)→FeBr2(aq)+H2(g)
What mass of HBr (in g) would you need to dissolve a 2.8-g pure iron bar on a padlock?
What mass of H2 would be produced by the complete reaction of the iron bar?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:00
Which statement best explains the relationship between an area is geography and the temperature of its surface water
Answers: 1

Chemistry, 22.06.2019 12:30
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3

Chemistry, 22.06.2019 15:00
‘which reaction would most likely require the use of an inert electrode?
Answers: 1

Chemistry, 22.06.2019 17:20
Pegmatites are igneous rocks in which the individual minerals are very large. typically, the minerals are all light-colored quartz, feldspar and muscovite. if you were given a black and white photograph of a pegmatite in a quarry (where the rock has been blasted and broken), what physical properties could you use to identify those three minerals in this hypothetical photo? describe each mineral and the specific diagnostic properties. be specific.
Answers: 2
You know the right answer?
Hydrobromic acid dissolves solid iron according to the following reaction:
Fe(s)+2HBr(aq)→FeBr2(aq...
Questions

Mathematics, 24.04.2020 01:04

Mathematics, 24.04.2020 01:04

History, 24.04.2020 01:04




Biology, 24.04.2020 01:04

History, 24.04.2020 01:04

Mathematics, 24.04.2020 01:04

Chemistry, 24.04.2020 01:04


Mathematics, 24.04.2020 01:04




Mathematics, 24.04.2020 01:04

History, 24.04.2020 01:04



Mathematics, 24.04.2020 01:05

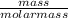
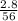 = 0.05mole
= 0.05mole 

