
Chemistry, 17.01.2021 19:10 EMscary4996
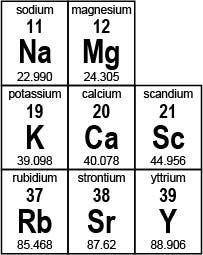
Use the portion of the periodic table shown below to answer the questions.
A portion of the first three columns of the periodic table is shown. Column one from top to bottom reads 11 sodium 22.990, 19 potassium 39.098, and 37 rubidium 85.468. Column two reads 12 magnesium 24.305, 20 calcium 40.078, and 38 strontium 87.62. Column three reads 21 scandium 44.956 and 39 yttrium 88.906.
Part 1: Name two elements that have the same properties as potassium (K). (2 points)
Part 2: Determine the number of protons, electrons, and neutrons present in an atom of magnesium (Mg). Explain how you determined your answer using complete sentences. (6 points)

Answers: 3
Another question on Chemistry


Chemistry, 21.06.2019 22:30
Each of the following compounds contains a metal that can exhibit more than one ionic charge. provide systematic names for each of these compounds. (a) cr(clo3)6 (b) mo(cn)6 (c) cr2(so3)3 (d) v(clo2)2 (e) v(cn)5 (f) os(clo2)4
Answers: 3

Chemistry, 22.06.2019 05:00
Agas can holds 2.0 gal of gasoline. what is this quantity in cubic centimeters?
Answers: 2

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1
You know the right answer?
Use the portion of the periodic table shown below to answer the questions.
A portion of the first t...
Questions

Social Studies, 30.07.2019 06:50

Mathematics, 30.07.2019 06:50


Social Studies, 30.07.2019 06:50



Mathematics, 30.07.2019 06:50




English, 30.07.2019 06:50



Computers and Technology, 30.07.2019 06:50


Computers and Technology, 30.07.2019 06:50



English, 30.07.2019 06:50

Mathematics, 30.07.2019 06:50



