
Chemistry, 17.01.2021 07:50 littlemrslazy
Consider the reaction 4FeS2 + 11O2 → 2Fe2O3 + 8SO2.
If 8 moles of FeS2 react with 15 moles of O2, what is the limiting reactant?
FeS_2
Fe_2O_3
O_2
SO_2
(The underscores with the number represents the number being subscripted)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1

Chemistry, 22.06.2019 19:50
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1

Chemistry, 23.06.2019 01:30
What is the importance of interlocking the fingers and rubbing while washing hands? the palms are the dirtiest parts of the hands. the spaces between the fingers get washed. the backs of the hands get washed. the fingernails are the dirtiest parts of the hands
Answers: 1

Chemistry, 23.06.2019 08:30
Which can be observed only in a microscopic view? a) structure of a muscle cell b) shape of a soybean plant c) foam insulation d) x-ray of a knee joint
Answers: 2
You know the right answer?
Consider the reaction 4FeS2 + 11O2 → 2Fe2O3 + 8SO2.
If 8 moles of FeS2 react with 15 moles of O2, w...
Questions


Mathematics, 01.06.2021 04:10

Mathematics, 01.06.2021 04:10

Physics, 01.06.2021 04:10



Mathematics, 01.06.2021 04:10



German, 01.06.2021 04:10

Mathematics, 01.06.2021 04:10




Mathematics, 01.06.2021 04:10

Social Studies, 01.06.2021 04:10




Physics, 01.06.2021 04:10

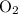 .
. 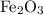 nor
nor 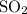 are on the right-hand side of the equation; these two species are products, not reactants. The only two reactants are
are on the right-hand side of the equation; these two species are products, not reactants. The only two reactants are 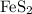 and
and 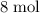 of its formula units are consumed. That is:
of its formula units are consumed. That is: 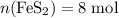 .
.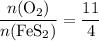 .
.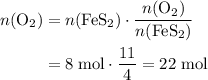 .
.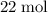 of
of 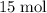 of
of 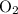 was available. Therefore, some
was available. Therefore, some 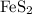 formula units would not have been consumed;
formula units would not have been consumed; 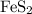 would be the reactant in excess, whereas
would be the reactant in excess, whereas 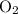 would be the limiting reactant.
would be the limiting reactant.

