Chemistry, 17.01.2021 06:30 johnplog2924
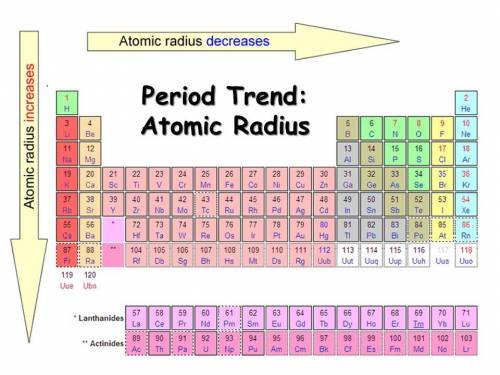
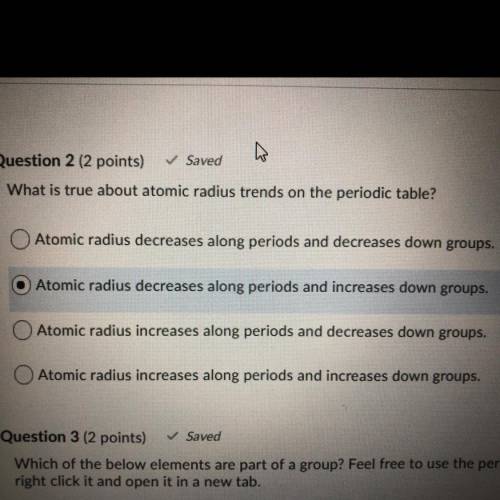
What is true about atomic radius trends on the periodic table?
A. Atomic radius decreases along periods and decreases down groups.
B. Atomic radius decreases along periods and increases down groups.
C. Atomic radius increases along periods and decreases down groups.
D. Atomic radius increases along periods and increases down groups.
It is not b

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1

Chemistry, 22.06.2019 12:30
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1

Chemistry, 22.06.2019 16:00
How could a student test the effect of removing heat from a gas that is stored in a sealed container? what must occur in order for matter to change states?
Answers: 2
You know the right answer?
What is true about atomic radius trends on the periodic table?
A. Atomic radius decreases along per...
Questions








Spanish, 30.10.2020 17:40

Mathematics, 30.10.2020 17:40

Biology, 30.10.2020 17:40


Mathematics, 30.10.2020 17:40

Mathematics, 30.10.2020 17:40


Mathematics, 30.10.2020 17:40

Mathematics, 30.10.2020 17:40

Arts, 30.10.2020 17:40

English, 30.10.2020 17:40