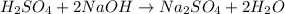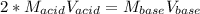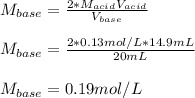Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Match the following items. 1. high-intensity bundle of energy being emitted from some decaying nuclei gamma ray 2. particle radiating from the nucleus of some atoms beta particle 3. negative particle identical to an electron but radiating from a decaying nucleus alpha particle
Answers: 1

Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium chlorate
Answers: 3

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 3
You know the right answer?
PLEASE HELP WHICH ONE IS IT AND CAN YOU EXPLAN HOW YOU GOT THE ANSWERS TY!!!
In a titration, a 20 m...
Questions

Mathematics, 09.12.2019 01:31


Biology, 09.12.2019 01:31

English, 09.12.2019 01:31

Spanish, 09.12.2019 01:31

Mathematics, 09.12.2019 01:31



Mathematics, 09.12.2019 01:31


Arts, 09.12.2019 01:31

Biology, 09.12.2019 01:31

History, 09.12.2019 01:31

Mathematics, 09.12.2019 01:31




Mathematics, 09.12.2019 02:31


Business, 09.12.2019 02:31