
Chemistry, 15.01.2021 18:20 sciencefanfae7248
Data
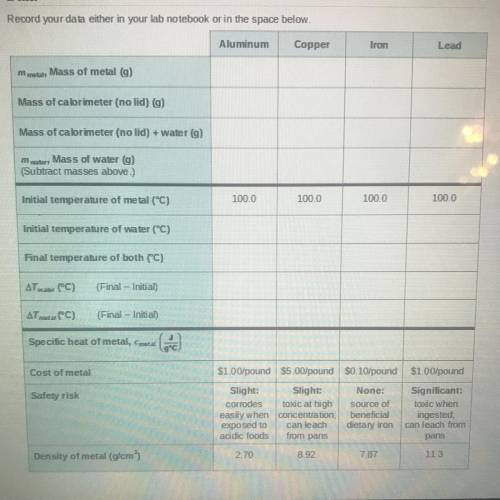
Record your data either in your lab notebook or in the space below.
Aluminum
Copper
Iron
Lead
m
mobile
Mass of metal g)
Mass of calorimeter (no lid) g)
Mass of calorimeter (no lid) + water (g)
m
Mass of water g)
(Subtract masses above.)
100.0
100.0
100.0
Initial temperature of metal (°C)
100.0
Initial temperature of water (°C)
Final temperature of both (°C)
AT water (C)
(Final - Initial)
ATmetal (°C)
(Final - Initial)
Specific heat of metal, Cmetal

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:50
Which of the following electromagnetic waves can create ions?
Answers: 2

Chemistry, 22.06.2019 11:50
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 22.06.2019 15:00
How is the shape of the poem “peer” connected to its meaning?
Answers: 2

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2
You know the right answer?
Data
Record your data either in your lab notebook or in the space below.
Aluminum
Coppe...
Aluminum
Coppe...
Questions


Biology, 09.09.2021 21:40


Computers and Technology, 09.09.2021 21:40


Mathematics, 09.09.2021 21:40

Mathematics, 09.09.2021 21:40

History, 09.09.2021 21:40



Mathematics, 09.09.2021 21:40

Mathematics, 09.09.2021 21:40





Mathematics, 09.09.2021 21:40


Mathematics, 09.09.2021 21:40

Mathematics, 09.09.2021 21:40



