
Chemistry, 14.01.2021 23:00 clashofclans17
50 POINTS PLEASE NO FAKE ANSWERS I REALLY NEED THESE ANSWERED
1. The following reaction shows calcium chloride reacting with silver nitrate.
CaCl2 + 2AgNO3 → 2AgCl + Ca(NO3)2
How many grams of AgCl are produced from 30.0 grams of CaCl2?
(Molar mass of Ca = 40.078 g/mol, Cl = 35.453 g/mol, O = 15.999 g/mol, Ag = 107.868 g/mol, N = 14.007 g/mol)
19.4 grams
38.8 grams
58.2 grams
77.5 grams
2. The table shows the recipe and the available ingredients for making the maximum possible number of sandwiches.
Making Sandwiches
Recipe for One Sandwich Ingredients Available
2 cheese slices, 1 ham slice, 2 bread slices 12 cheese slices, 10 ham slices, 12 bread slices
If the ingredients represent reactants of a chemical reaction, which of the following represents the leftover reactant?
Two ham slices
Four ham slices
Two cheese slices
Four cheese slices
3. Read the given chemical reaction.
C2H6 + O2 → CO2 + H2O
How many moles of H2O are produced during the complete combustion of 1.4 moles of C2H6?
2.8 moles
4.2 moles
5.6 moles
7.0 moles
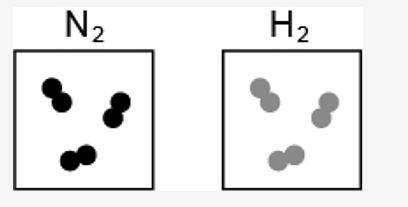
4. The image represents the reaction between a certain number of molecules of N2 and H2.
[IMAGE INCLUDED]
If the maximum possible amount of NH3 is formed during the reaction, what is the leftover reactant?
One molecule of N2
One molecule of H2
Two molecules of N2
Two molecules of H2

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Agas at 155 kpa and standard temperature has an initial volume of 1.00 l. the pressure of the gas rises to 500 kpa as the temperature also rises to 135°c. what is the new volume? 2.16 l 0.463 l 0.207 l 4.82 l
Answers: 3

Chemistry, 22.06.2019 00:00
1) these are barrel shaped microtubules in most animal cells, that organize the spindles during cell division
Answers: 1

Chemistry, 22.06.2019 07:30
Label a-f based on the table using c for concentrated and d for dilute
Answers: 2

You know the right answer?
50 POINTS PLEASE NO FAKE ANSWERS I REALLY NEED THESE ANSWERED
1. The following reaction shows calci...
Questions

Physics, 31.08.2019 20:20

History, 31.08.2019 20:20

History, 31.08.2019 20:20


Mathematics, 31.08.2019 20:20


Biology, 31.08.2019 20:20

Mathematics, 31.08.2019 20:20

Chemistry, 31.08.2019 20:20

Business, 31.08.2019 20:20

Biology, 31.08.2019 20:20



Health, 31.08.2019 20:20




Computers and Technology, 31.08.2019 20:20

Spanish, 31.08.2019 20:20

Computers and Technology, 31.08.2019 20:20



