
Chemistry, 13.01.2021 15:40 olivya2005d
If you had excess chlorine, how many moles of of aluminum chloride could be produced from 11.0 g of aluminum

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 08:30
Since the gas in your graduated cylinder is a mixture of butane and water vapor, you must determine the partial pressure of the butane, pbutane, alone. to do this, consult a reference and record the partial pressure of the water vapor, pwater, at the temperature you recorded. use the following formula to compute the partial pressure of the butane. pbutane = atmosphere - pwater use the following combined gas law formula and compute the volume that the butane sample will occupy at stp. (hint: convert both temperatures to kelvin.) pbutane x voriginal = pstandard x vfinal troom tstandard use the following ratio and proportion formula to determine the mass of butane needed to occupy a volume of 22.4 l at stp. grams of butane you used “x” grams of butane ml of butane corrected to stp = 22,400 ml compute the theoretical molar mass of butane based on its formula and the atomic masses on the periodic table. compare your experimental results from #3 to the theoretical value of #4, computing a percent error of your findings using this formula: % error = measured value - accepted value x 100 accepted value use the following ratio and proportion formula to determine the mass of butane needed to occupy a volume of 22.4 l at stp. need asap
Answers: 1

Chemistry, 22.06.2019 10:30
Earth's axis of rotation is tilted at an angle of 23.5 degrees. what is one change you would see on earth if its axis was not tilted?
Answers: 3

Chemistry, 22.06.2019 21:30
What is another way to determine mass times acceleration?
Answers: 1

Chemistry, 22.06.2019 21:30
Which of the following changes will decrease the total amount of gaseous solute able to be dissolved in a liter of liquid water? (2 points) decreasing temperature decreasing pressure decreasing surface area decreasing solute concentration
Answers: 1
You know the right answer?
If you had excess chlorine, how many moles of of aluminum chloride could be produced from 11.0 g of...
Questions


Mathematics, 28.09.2019 07:00

Social Studies, 28.09.2019 07:00


Health, 28.09.2019 07:00

Mathematics, 28.09.2019 07:00

Biology, 28.09.2019 07:00


History, 28.09.2019 07:00

Physics, 28.09.2019 07:00


Mathematics, 28.09.2019 07:00


Social Studies, 28.09.2019 07:00

Arts, 28.09.2019 07:00


English, 28.09.2019 07:00

English, 28.09.2019 07:00

French, 28.09.2019 07:00

Computers and Technology, 28.09.2019 07:00

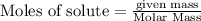
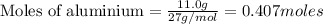
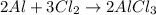
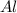 is the limiting reagent as it limits the formation of product and
is the limiting reagent as it limits the formation of product and 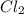 is the excess reagent.
is the excess reagent.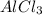
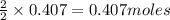 of
of 

