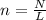Chemistry, 10.01.2021 23:50 joannamarquez0701
How many moles of carbon are in 7.00 x 1023 atoms of carbon?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 18:50
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1

Chemistry, 22.06.2019 22:30
Gusing the milligrams of ascorbic acid you entered above, the ratio of total sample volume to aliquot volume, and the total milligrams of the vitamin c tablet that you dissolved, calculate the mass of ascorbic acid in the vitamin c tablet for each trial. do this by scaling up to find the amount (mg) of ascorbic acid in your 250 ml flask. enter your calculated mass of ascorbic acid in the vitamin c tablet, for each trial. be sure to enter your calculated mass in the corresponding order that you entered your milligrams of ascorbic acid. the milligrams of ascorbic acid you entered for entry #1 previously should correspond to the mass of ascorbic acid that you enter for entry #1 here.
Answers: 1

Chemistry, 23.06.2019 02:30
When the ionic compound nabr dissolves in water, br– ions are pulled into solution by the attraction between what two particles? a. the na+ and br– ions b. the na+ ion and the negative end of a water molecule c. the br– ion and the positive end of a water molecule d. the br– ion and the negative end of a water molecule
Answers: 1
You know the right answer?
How many moles of carbon are in 7.00 x 1023 atoms of carbon?...
Questions

Mathematics, 26.05.2020 20:59







Mathematics, 26.05.2020 20:59

French, 26.05.2020 20:59

Mathematics, 26.05.2020 20:59



History, 26.05.2020 20:59

Mathematics, 26.05.2020 20:59

Chemistry, 26.05.2020 20:59




Mathematics, 26.05.2020 20:59

Mathematics, 26.05.2020 20:59