
Chemistry, 10.01.2021 22:20 Itsyourgirllulu
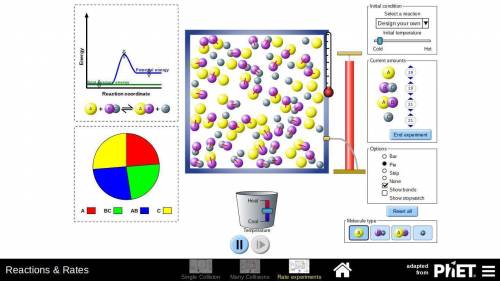
In part D of task 1, you identified at least two ways in which the reaction of nitrogen and hydrogen could be changed to alter the equilibrium. Use the simulation to test those changes. Describe how you used the simulation to model the changes and the results they produced. Use these methods if you find them helpful:
Look at the pie graph to see how the system changes.
Use the Temperature slider at the bottom to cool or heat the mixture.
Click the pause button on the simulation to observe the number of particles at any point of time.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:00
Use examples from the article to explain one positive and one negative effect that chemistry has had on society
Answers: 2

Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1


Chemistry, 23.06.2019 01:30
Some molecular compounds, such as hcl, ionize completely in solution. for molecular compounds such as h2co3, most molecules do not ionize in solution. which describes the properties of these two solutes? a. hcl and h2co3 have the same effect on the properties of the solution. b. hcl raises the freezing point of water more than h2co3 does. c. hcl raises the boiling point of water more than h2co3 does.
Answers: 2
You know the right answer?
In part D of task 1, you identified at least two ways in which the reaction of nitrogen and hydrogen...
Questions

English, 31.01.2021 01:00


Mathematics, 31.01.2021 01:00

Arts, 31.01.2021 01:00

Mathematics, 31.01.2021 01:00





Mathematics, 31.01.2021 01:00

Mathematics, 31.01.2021 01:00




Mathematics, 31.01.2021 01:00



Biology, 31.01.2021 01:00




