
Nitrogen dioxide can be made with the reaction of nitrogen monoxide and oxygen.
Assuming that the reaction is at equilibrium, what effect would a decrease in the concentration of NO_2 have on the equilibrium position once the equilibrium is reestablished?
A. An increase in the amount of begin mathsize 14px style NO subscript 2 end style.
B. An increase in the amount of begin mathsize 14px style straight O subscript 2 end style.
C. No change in the amount of begin mathsize 14px style straight O subscript 2 end style.
D. An increase in the amount of NO.
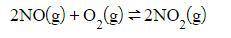

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 04:30
Use the periodic table to determine the electron configuration of dysprosium (dy) and americium (am) in noble-gas notation.
Answers: 1

Chemistry, 22.06.2019 08:30
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3

Chemistry, 22.06.2019 15:20
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3
You know the right answer?
Nitrogen dioxide can be made with the reaction of nitrogen monoxide and oxygen.
Assuming that the r...
Questions

Biology, 01.04.2021 22:40

Mathematics, 01.04.2021 22:40

Mathematics, 01.04.2021 22:40

English, 01.04.2021 22:40

English, 01.04.2021 22:40

English, 01.04.2021 22:40


History, 01.04.2021 22:40

Arts, 01.04.2021 22:40

Biology, 01.04.2021 22:40

History, 01.04.2021 22:40

Biology, 01.04.2021 22:40

Mathematics, 01.04.2021 22:40


Mathematics, 01.04.2021 22:40

Mathematics, 01.04.2021 22:40

English, 01.04.2021 22:40


Mathematics, 01.04.2021 22:40

Mathematics, 01.04.2021 22:40



