
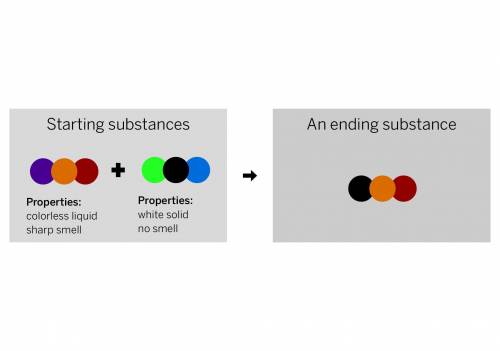
A chemist mixed two substances together: a colorless liquid with a strong smell and a white solid with no smell. The substances’ repeating groups of atoms are shown above on the left. After they were mixed, the chemist analyzed the results and found two substances. One ending substance had the repeating group of atoms shown above on the right. Is the ending substance the same substance as the colorless liquid? What happened to the atoms of the starting substances when the ending substances formed? Be sure to explain your answers to both of these questions.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 12:40
Consider the directing effects of the substituents on salicylamide and predict the possible structures of the iodination products. which do you think will be the major product?
Answers: 1

Chemistry, 22.06.2019 15:30
How does a large body of water, such as the ocean, influence climate?
Answers: 1

Chemistry, 22.06.2019 20:00
Iam hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 1
You know the right answer?
A chemist mixed two substances together: a colorless liquid with a strong smell and a white solid wi...
Questions

Advanced Placement (AP), 15.06.2021 05:30






History, 15.06.2021 05:30

Mathematics, 15.06.2021 05:30



Mathematics, 15.06.2021 05:30


History, 15.06.2021 05:30

Mathematics, 15.06.2021 05:30

Mathematics, 15.06.2021 05:30

Mathematics, 15.06.2021 05:30






