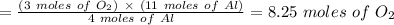Chemistry, 08.01.2021 18:40 maytce7237
Using the balanced chemical equation below. 2Al2O3 --> 4Al + 3O2 How many moles of oxygen are produced if 11.0 mol of Al are produced?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 22.06.2019 17:30
Upon decomposition, one sample of magnesium fluoride produced 1.65 kg of magnesium and 2.56 kg of fluorine. a second sample produced 1.32 kg of magnesium. part a how much fluorine (in grams) did the second sample produce?
Answers: 2

Chemistry, 22.06.2019 19:30
Anurse used a 0.02-mg/l solution of disinfection to clean a patients wound. what is the concentration of the solution expressed as a percentage?
Answers: 1
You know the right answer?
Using the balanced chemical equation below. 2Al2O3 --> 4Al + 3O2 How many moles of oxygen are pro...
Questions

Mathematics, 12.04.2021 03:40



Mathematics, 12.04.2021 03:40

Mathematics, 12.04.2021 03:40

Social Studies, 12.04.2021 03:40

Mathematics, 12.04.2021 03:40

Advanced Placement (AP), 12.04.2021 03:40

Mathematics, 12.04.2021 03:40






English, 12.04.2021 03:40



History, 12.04.2021 03:40

Mathematics, 12.04.2021 03:40