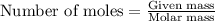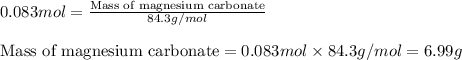3) (Solid magnesium carbonate mixed in a solution of dilute hydrochloric acid. Assume the
specific heat capacity of the solution is 4.18 J/g°C. The enthalpy of the reaction is -23.4
kJ. If 1950 J of heat are released by this reaction, how many grams of Mgco, were
dissolved?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:10
Why is the vapor pressure of a warm lake higher than the vapor pressure of a cold lake? o a. warm water has a greater heat of vaporization. ob. warm water evaporates more quickly. cool water evaporates more quickly. od. cool water has a greater heat of vaporization.
Answers: 1

Chemistry, 21.06.2019 20:30
Five students had to answer the question how are elements arranged in a periodic tabledamon: i think the elements are arranged by increasing massflo: i think the elements are arranged according to their properties sienna: i think the elements are arranged by when their discovers kyle: i think the elements are arranged according to how common they areglenda: i don't agree with any of themwho is right
Answers: 1


Chemistry, 22.06.2019 10:20
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3
You know the right answer?
3) (Solid magnesium carbonate mixed in a solution of dilute hydrochloric acid. Assume the
specific...
Questions







Engineering, 24.11.2020 14:00


English, 24.11.2020 14:00

Advanced Placement (AP), 24.11.2020 14:00

English, 24.11.2020 14:00

Chemistry, 24.11.2020 14:00

Mathematics, 24.11.2020 14:00


Mathematics, 24.11.2020 14:00

Computers and Technology, 24.11.2020 14:00

Mathematics, 24.11.2020 14:00


Geography, 24.11.2020 14:00

Biology, 24.11.2020 14:00

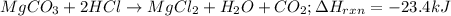
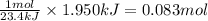 of magnesium carbonate is dissolved
of magnesium carbonate is dissolved