
Chemistry, 07.01.2021 23:50 gshreya2005
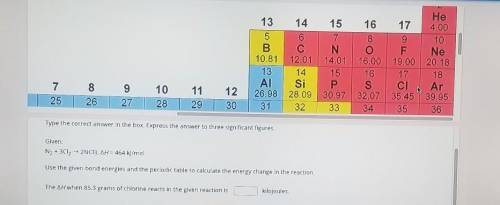
Type the correct answer in the box, Express the answer to three significant figures. Given: N2 + 3Cl2 + 2NC3, AH = 464 kJ/mol Use the given bond energies and the periodic table to calculate the energy change in the reaction. The AH when 85.3 grams of chlorine reacts in the given reaction is kilojoules.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Write the empirical chemical formula of calcium with a mass percent of 38.8, phosphorus with a mass percent of 20.0, and oxygen with a mass percent of 41.3.
Answers: 1

Chemistry, 23.06.2019 05:00
If 15 drops of ethanol from a medicine dropper weigh 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? density of ethanol is ethanol is 0.80g/ml.
Answers: 2

Chemistry, 23.06.2019 06:40
A250 g sample of water with an initial temperatureof 98.8 closes 6500 joules of heat. what is the finaltemperature of the water?
Answers: 1

Chemistry, 23.06.2019 08:30
According to the passage, which of these is true about gray water systems? a) gray water systems use plants that require less water. eliminate b) gray water systems require the use of less fossil fuels. c) gray water systems reduce the amount of fresh water used. d) gray water systems reduce the amount water used by shower heads.
Answers: 1
You know the right answer?
Type the correct answer in the box, Express the answer to three significant figures. Given: N2 + 3Cl...
Questions


English, 11.10.2019 19:10

Physics, 11.10.2019 19:10

Physics, 11.10.2019 19:10


Mathematics, 11.10.2019 19:10

Mathematics, 11.10.2019 19:10

English, 11.10.2019 19:10




Social Studies, 11.10.2019 19:10


Biology, 11.10.2019 19:10








