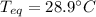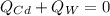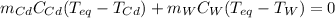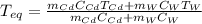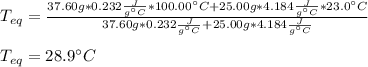Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1


Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1
You know the right answer?
If a piece of cadmium with a mass of 37.60 g and a temperature of 100.0 oC is dropped into 25.00 cc...
Questions




Biology, 31.07.2019 14:30









Mathematics, 31.07.2019 14:30

Biology, 31.07.2019 14:30





History, 31.07.2019 14:30

Business, 31.07.2019 14:30