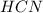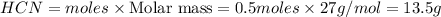Methane (CH4), ammonia (NH3), and oxygen (O2) can react to form hydrogen cyanide (HCN) and water according to this equation:
CH4+NH3+O2→HCN+H2O. A student has 8 g of methane and 10 g of ammonia in excess oxygen.
a. What is the balanced equation for this reaction?
b. Which reagent is limiting? Explain why.
c. How many grams of hydrogen cyanide will be formed?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:30
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1

Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3

Chemistry, 23.06.2019 01:30
Ariver current has a velocity of 5km/h relative to the shore, and a boat moves in the same direction as the current at 5 km/h relative to the river. how can the velocity of the boat relative to the shore be calculated?
Answers: 1

Chemistry, 23.06.2019 01:30
The solubility of barium nitrate is 9.02 g/100 g h2o at 20°c. a 15.2 g sample of barium nitrate is added to 200.0 g of water at 20°c. is the solution saturated, unsaturated, or supersaturated? a. unsaturated b. saturated c. supersaturated
Answers: 1
You know the right answer?
Methane (CH4), ammonia (NH3), and oxygen (O2) can react to form hydrogen cyanide (HCN) and water acc...
Questions


Mathematics, 10.06.2021 08:50




History, 10.06.2021 08:50


Computers and Technology, 10.06.2021 08:50

Mathematics, 10.06.2021 08:50

Mathematics, 10.06.2021 08:50


History, 10.06.2021 08:50


English, 10.06.2021 08:50


Mathematics, 10.06.2021 08:50

Mathematics, 10.06.2021 09:00

English, 10.06.2021 09:00

Mathematics, 10.06.2021 09:00

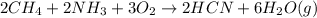
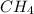 is the limiting.
is the limiting.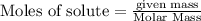
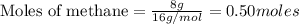
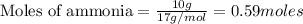
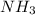
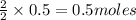 of
of