
1. The gases SO2, O2 and SO3 are allowed to reach equilibrium at a constant temperature. The equilibrium constant for the reaction
2SO2(g) + O2(g) -> 2S03(g)
is 1.6 x 104 atm-1
a) Calculate the value of Kp for the reaction
SO2(g) + ½O2(g) -> SO3(g)
(b) The equilibrium constant for the dissociation of Pcl5(g) to form PCl3(g) and Cl2(g) is
0.04 at 250°C. An equilibrium mixture contains 0.20 mol PC13 and 0.12 mol Cl, in a
4000 cm container.
i) Write the chemical equation.
ii) Calculate the concentration of PCIs in this container.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 14:10
Precision can be defined as the o exact center of a data set. o reproducibility of a measured value. o correlation between two variables that are measured in a data set agreement between a measured value and an accepted value.
Answers: 2


Chemistry, 22.06.2019 22:00
The volume of an unknown substance in a sealed glass jar is 50 milliliters. the volume of the jar is 200 milliliters. which state of matter could the substance be?
Answers: 2
You know the right answer?
1. The gases SO2, O2 and SO3 are allowed to reach equilibrium at a constant temperature. The equilib...
Questions


Mathematics, 26.03.2020 20:03

Mathematics, 26.03.2020 20:03

History, 26.03.2020 20:03

Social Studies, 26.03.2020 20:03


English, 26.03.2020 20:03





Mathematics, 26.03.2020 20:03




Mathematics, 26.03.2020 20:03


English, 26.03.2020 20:03


Mathematics, 26.03.2020 20:03

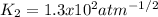
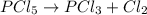
![[PCl_5]=0.0375M](/tpl/images/1006/3442/49c8a.png)
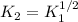
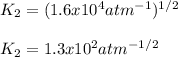
![K=\frac{[PCl_3][Cl_2]}{[PCl_5]}](/tpl/images/1006/3442/bc353.png)
![[PCl_5]=\frac{[PCl_3][Cl_2]}{K}\\](/tpl/images/1006/3442/edc14.png)
![[PCl_5]=\frac{\frac{0.20mol}{4L} *\frac{0.12mol}{4L} }{0.04}](/tpl/images/1006/3442/09a24.png)


