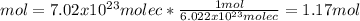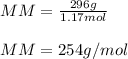Chemistry, 22.12.2020 04:20 dchannakhone84
7.02 x 10^23 molecules of X2, a ssubstance consisting of diatomic molecules, has a mass of 296 grams. Determine the atomic weight (mass of a mole of atoms) of element X.
a. 254 g/mol.
b. 296 g/mol.
c. 148 g/mol.
d. 127 g/mol.
e. 507 g/mol.

Answers: 2
Another question on Chemistry



Chemistry, 22.06.2019 06:00
If you burn 10 kilograms of wood in a fire (combustion) what is the weight of the products after the fire has finished burning the wood?
Answers: 3

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1
You know the right answer?
7.02 x 10^23 molecules of X2, a ssubstance consisting of diatomic molecules, has a mass of 296 grams...
Questions





Computers and Technology, 19.11.2019 20:31

Mathematics, 19.11.2019 20:31





Computers and Technology, 19.11.2019 20:31





Biology, 19.11.2019 20:31