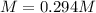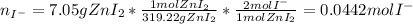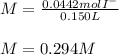Chemistry, 21.12.2020 17:30 aidenbender06
Suppose 7.05 g of zinc iodide is dissolved in 150. mL of a 0.20M aqueous solution of potassium carbonate. Calculate the final molarity of iodide anion in the solution. You can assume the volume of the solution doesn't change when the zinc iodide is dissolved in it. Round your answer to 3 significant digits.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 14:30
The three types is stress that act on earths rocks are compression, tension, and
Answers: 1

Chemistry, 22.06.2019 22:30
What if it is did darwin used to support his theory of evolution
Answers: 1


Chemistry, 23.06.2019 03:00
Abaker touches a pie right after taking it out of the oven. which statement best explains why the pie feels hot?
Answers: 2
You know the right answer?
Suppose 7.05 g of zinc iodide is dissolved in 150. mL of a 0.20M aqueous solution of potassium carbo...
Questions

Mathematics, 24.06.2019 11:00

Social Studies, 24.06.2019 11:00







Mathematics, 24.06.2019 11:00


Mathematics, 24.06.2019 11:00

Geography, 24.06.2019 11:00

English, 24.06.2019 11:00

Biology, 24.06.2019 11:00

History, 24.06.2019 11:00

History, 24.06.2019 11:00


Chemistry, 24.06.2019 11:00


Mathematics, 24.06.2019 11:00