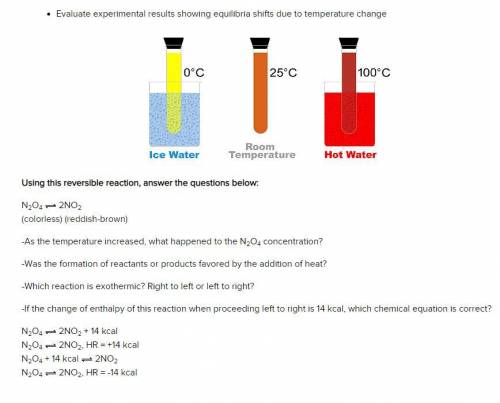
Using this reversible reaction, answer the questions below:
N2O4 2NO2
(colorless) (reddish-br...

Chemistry, 21.12.2020 14:00 mommer2019
Using this reversible reaction, answer the questions below:
N2O4 2NO2
(colorless) (reddish-brown)
-As the temperature increased, what happened to the N2O4 concentration?
-Was the formation of reactants or products favored by the addition of heat?
-Which reaction is exothermic? Right to left or left to right?
-If the change of enthalpy of this reaction when proceeding left to right is 14 kcal, which chemical equation is correct?
N2O4 2NO2 + 14 kcal
N2O4 2NO2, HR = +14 kcal
N2O4 + 14 kcal 2NO2
N2O4 2NO2, HR = -14 kcal

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 09:00
Astudent is asked to identify and element that is pale yellow brittle solid and does not conduct electricity. at which location in this periodic table would the element most likely be found?
Answers: 2


Chemistry, 22.06.2019 22:30
What is the work done by the electric force to move a 1 c charge from a to b?
Answers: 2
You know the right answer?
Questions

Mathematics, 26.07.2021 16:30



Mathematics, 26.07.2021 16:30





Social Studies, 26.07.2021 16:30

Mathematics, 26.07.2021 16:30

Social Studies, 26.07.2021 16:30

Mathematics, 26.07.2021 16:30


Social Studies, 26.07.2021 16:30


Chemistry, 26.07.2021 16:30

Computers and Technology, 26.07.2021 16:30


Biology, 26.07.2021 16:30




