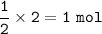Based on the following balanced equation:
H2 + O2 à H2O
How many moles of O2 are required to...

Chemistry, 20.12.2020 09:00 igsgymnast
Based on the following balanced equation:
H2 + O2 à H2O
How many moles of O2 are required to react completely with 2.00 moles of H2

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3

Chemistry, 22.06.2019 04:30
Why are people not able to scuba dive in the deep part of the ocean
Answers: 2

Chemistry, 22.06.2019 05:50
What are transitions between a liquid and a solid called? identify which way they are transitioning
Answers: 2

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3
You know the right answer?
Questions

History, 05.05.2020 06:01

Mathematics, 05.05.2020 06:01

English, 05.05.2020 06:01

English, 05.05.2020 06:01


Chemistry, 05.05.2020 06:01




Mathematics, 05.05.2020 06:01



Mathematics, 05.05.2020 06:01





Mathematics, 05.05.2020 06:01