Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:50
Blank allows you to do calculations for situations in which only the amount of gas is constant a)boyle's law b)combined gas law c)ideal gas law d)dalton's law
Answers: 1


Chemistry, 22.06.2019 10:50
A100 kmol/h stream that is 97 mole% carbon tetrachloride (ccl4) and 3% carbon disulfide (cs2) is to be recovered from the bottom of a distillation column. the feed to the column is 16 mole% cs2 and 84% ccl4, and 2% of the ccl4 entering the column is contained in the overhead stream leaving the top of the column. calculate the mass and mole fractions of ccl4 in the overhead stream, and determine the molar flow rates of ccl4 and cs2 in the overhead and feed streams. 12. mw_ccla- 153.82; mw_cs2-76.14.
Answers: 3

Chemistry, 22.06.2019 12:30
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3
You know the right answer?
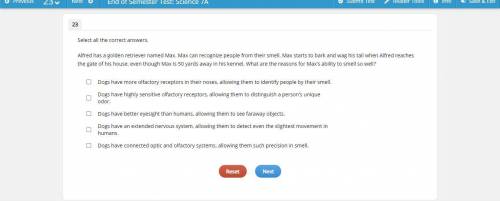
Plz help me.I need 2 answers
...
...
Questions



Physics, 11.10.2020 14:01






Mathematics, 11.10.2020 14:01

Spanish, 11.10.2020 14:01



Mathematics, 11.10.2020 14:01


Mathematics, 11.10.2020 14:01

Physics, 11.10.2020 14:01


Geography, 11.10.2020 14:01

Social Studies, 11.10.2020 14:01

Biology, 11.10.2020 14:01