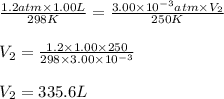Chemistry, 18.12.2020 17:40 Roselyn0131
A gas-filled balloon having a volume of 1.00 L at 1.2 atm and 25°C is allowed to rise to the stratosphere (about 30 km above the surface of Earth), where the temperature and pressure are -23°C and 3.00 10-3 atm, respectively. Calculate the final volume of the balloon.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 14:00
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1


Chemistry, 23.06.2019 05:30
How many moles are in 1.26*10^24 particles in significant figures
Answers: 2

Chemistry, 23.06.2019 15:30
Which answer below correctly identifies the type of change and the explanation when magnesium comes into contact with hydrochloric acid
Answers: 1
You know the right answer?
A gas-filled balloon having a volume of 1.00 L at 1.2 atm and 25°C is allowed to rise to the stratos...
Questions


Biology, 23.04.2020 21:48









Mathematics, 23.04.2020 21:48

History, 23.04.2020 21:48

Engineering, 23.04.2020 21:48

Chemistry, 23.04.2020 21:48

Advanced Placement (AP), 23.04.2020 21:48

Mathematics, 23.04.2020 21:48


World Languages, 23.04.2020 21:48


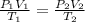
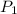 = Initial pressure = 1.2 atm
= Initial pressure = 1.2 atm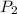 = Final pressure =
= Final pressure = 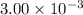 atm
atm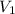 = Initial volume = 1.00 L
= Initial volume = 1.00 L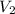 = Final volume = ?
= Final volume = ?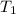 = Initial temperature =
= Initial temperature = ![25^oC=[25+273]=298K](/tpl/images/0999/9989/e58c7.png)
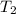 = Final temperature =
= Final temperature = ![-23^oC=[-23+273]=250K](/tpl/images/0999/9989/5a731.png)