
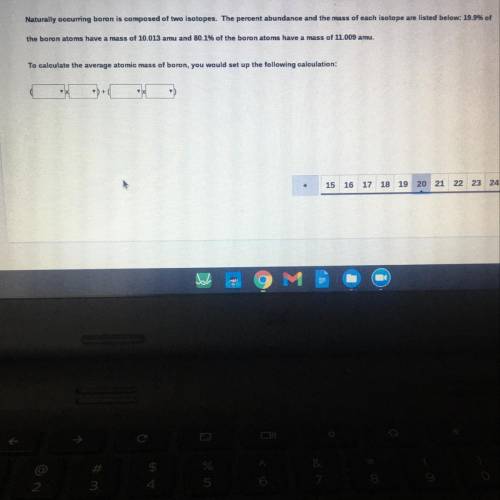
Naturally occurring boron is composed of two isotopes. The percent abundance and the mass of each isotope are listed below: 19.99
the boron atoms have a mass of 10.013 amu and 80.1% of the boron atoms have a mass of 11.009 amu.
To calculate the average atomic mass of boron, you would set up the following calculation:
+

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:40
Describe in detail the melting point behavior of the 80: 20 benzoic acid-mandelic acid mixture
Answers: 3

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2


Chemistry, 22.06.2019 21:30
How many liters of 3.0 m naoh solution will react with 0.60 liters of 4.0 m h2so4? h2so4 + naoh → na2so4 + h2o 1.2 l 1.6 l 2.4 l 2.8 l
Answers: 3
You know the right answer?
Naturally occurring boron is composed of two isotopes. The percent abundance and the mass of each is...
Questions

Mathematics, 24.11.2020 05:30


Social Studies, 24.11.2020 05:30


Mathematics, 24.11.2020 05:30

Advanced Placement (AP), 24.11.2020 05:40

Mathematics, 24.11.2020 05:40

Mathematics, 24.11.2020 05:40


Health, 24.11.2020 05:40

Engineering, 24.11.2020 05:40

Mathematics, 24.11.2020 05:40


English, 24.11.2020 05:40



English, 24.11.2020 05:40

Mathematics, 24.11.2020 05:40





