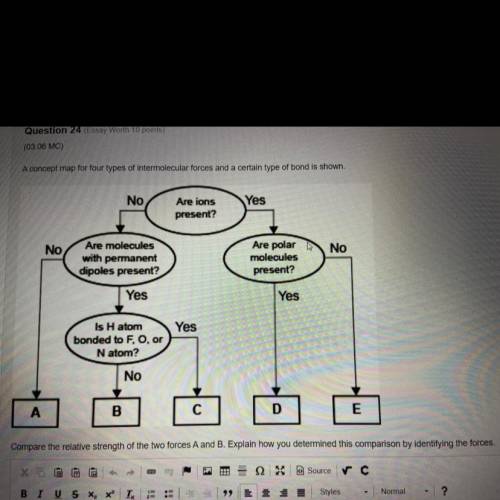
A concept map for four types of intermolecular forces and a certain type of bond is shown.
No
...

Chemistry, 17.12.2020 19:50 lizbethh62
A concept map for four types of intermolecular forces and a certain type of bond is shown.
No
Yes
Are ions
present?
No
No
Are molecules
with permanent
dipoles present?
Are polar
molecules
present?
Yes
Yes
Yes
Is H atom
bonded to F, O, or
Natom?
No
A
С
D
E
Compare the relative strength of the two forces A and B. Explain how you determined this comparison by identifying the forces.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 13:00
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 15:30
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3

Chemistry, 22.06.2019 22:30
Write and balance the chemical equation that represents the reaction of aqueous sulfuric acid with aqueous sodium hydroxide to form water and sodium sulfate. include phases.
Answers: 1
You know the right answer?
Questions


Mathematics, 04.07.2019 09:30

English, 04.07.2019 09:30

Chemistry, 04.07.2019 09:30


Mathematics, 04.07.2019 09:30


History, 04.07.2019 09:30



History, 04.07.2019 09:30

Mathematics, 04.07.2019 09:30





Mathematics, 04.07.2019 09:40


Mathematics, 04.07.2019 09:40

Physics, 04.07.2019 09:40



