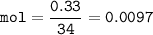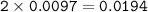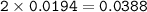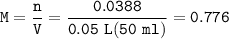Chemistry, 17.12.2020 02:30 getzperez1962
0.33 g of hydrosulfuric acid, H25, is added to 50.0 mL of water. What is the concentration (molarity) of [H301 of this solution?
H25 --> 2H +52
2H+ + 2 H20 --> 2 H30* + 2 OH
[Do not include units in your answer]

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 18:40
What is one real world example of a colligative property?
Answers: 2

Chemistry, 22.06.2019 20:50
What is the vapor pressure of a solution with a benzene to octane?
Answers: 2

Chemistry, 22.06.2019 23:00
How does the value of the equilibrium constant show that a reaction reaches equilibrium very quickly? (a) the equilibrium constant is large. (b) the equilibrium constant is small. (c) the equilibrium constant is zero. (d) the value of the equilibrium constant does not show how quickly a reaction comes to equilibrium.
Answers: 1
You know the right answer?
0.33 g of hydrosulfuric acid, H25, is added to 50.0 mL of water. What is the concentration (molarity...
Questions


Mathematics, 19.11.2020 08:20

Mathematics, 19.11.2020 08:20








Mathematics, 19.11.2020 08:20




Social Studies, 19.11.2020 08:20

Mathematics, 19.11.2020 08:20

Business, 19.11.2020 08:30

Mathematics, 19.11.2020 08:30

English, 19.11.2020 08:30

Mathematics, 19.11.2020 08:30