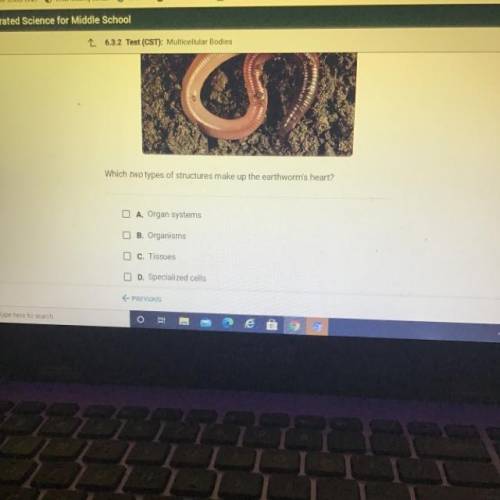
HELP ME PLEASE ILL MARK BRAINLEST
...

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:30
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 22.06.2019 15:30
Which suspect most likely committed the robbery and how do you know
Answers: 2

Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1

Chemistry, 22.06.2019 22:00
In order to complete this lab. you will need to be familiar with some common chemistry terms. complete the chemical change puzzle and list the relevant terms and their meaning below a.rectant b.product c.supernate
Answers: 3
You know the right answer?
Questions

English, 13.11.2020 21:50

Chemistry, 13.11.2020 21:50

Mathematics, 13.11.2020 21:50


Health, 13.11.2020 21:50


Mathematics, 13.11.2020 21:50

Biology, 13.11.2020 21:50

Social Studies, 13.11.2020 21:50

Mathematics, 13.11.2020 21:50

Mathematics, 13.11.2020 21:50



English, 13.11.2020 21:50


Mathematics, 13.11.2020 21:50




Mathematics, 13.11.2020 21:50