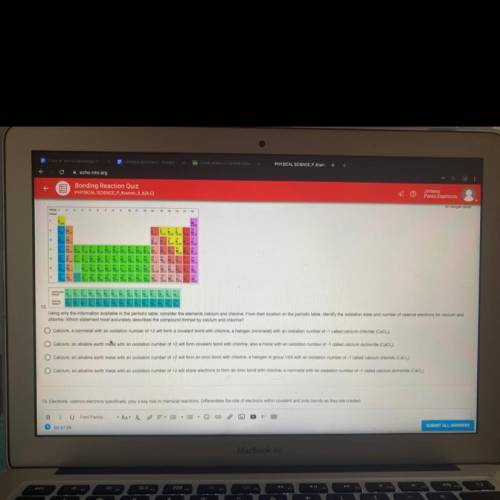
NEED HELP ON QUESTION 12 and 13 HELPP
...

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:20
What is the ima of the 1 st class lever in the graphic given? 2 3 0.5
Answers: 1

Chemistry, 22.06.2019 10:30
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1

Chemistry, 23.06.2019 09:00
The concentration of ionic substances is important for the heart to beat. your heart responds to electrical impulses that travel through heart cells that are made up mostly of water. which properties of ionic compounds are important to support this function? solubility in water conductivity crystalline melting point
Answers: 3

Chemistry, 23.06.2019 10:10
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10-3 m and k for the dissociation is 1.86x10-5. ch3cooh(aq)+h2o(l)+> h3o+(aq)+ch3coo-(aq) show me how to get the answer.
Answers: 3
You know the right answer?
Questions


Mathematics, 19.01.2021 21:10

Mathematics, 19.01.2021 21:10


Mathematics, 19.01.2021 21:10


English, 19.01.2021 21:10

Mathematics, 19.01.2021 21:10



Chemistry, 19.01.2021 21:10


Mathematics, 19.01.2021 21:10


Mathematics, 19.01.2021 21:10

Mathematics, 19.01.2021 21:10

Biology, 19.01.2021 21:10


Mathematics, 19.01.2021 21:10

Physics, 19.01.2021 21:10