
Chemistry, 15.12.2020 21:30 perezsamantha3oqr0za
You weigh out 43.9 g of potassium (K). How many moles of potassium did you weigh? How many atoms?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Fission of uranium-235 products energy and a. isotopes of smaller elements b. isotopes of larger elements c. lighter isotopes of uranium d. heavier isotopes of uranium
Answers: 3


Chemistry, 22.06.2019 16:00
Which of the following is the correct definition of chemical energy? a. energy an object has because of its motion or position b. energy resulting from the flow of charged particles, such as electrons or ions c. energy produced from the splitting of atoms d. energy stored in chemical bonds of molecules
Answers: 1

Chemistry, 23.06.2019 03:30
If you need to add 27.50ml of a solution, which piece of glassware would you use to deliver this volume and explain how you would determine if the 27.50 ml was measured?
Answers: 1
You know the right answer?
You weigh out 43.9 g of potassium (K). How many moles of potassium did you weigh? How many atoms?...
Questions


Mathematics, 13.03.2020 02:22


Computers and Technology, 13.03.2020 02:22

Mathematics, 13.03.2020 02:22




Mathematics, 13.03.2020 02:23








Mathematics, 13.03.2020 02:23


Mathematics, 13.03.2020 02:23

Mathematics, 13.03.2020 02:23

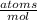 = 6.76x10²³ K atoms
= 6.76x10²³ K atoms

