
Chemistry, 15.12.2020 20:00 claytonp7695
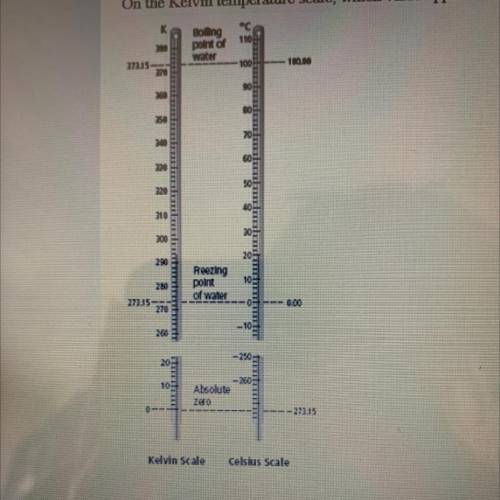
On the Kelvin temperature scale, which value approximately represents the freezing point of water?
Bolling
point of
water
1104
27215-
100
100.00
200
290
Reezing
point
of water
273.15-
0.00
Absolute
zero
273.15
Kelvin Scale
Celsius Scale
O A. 32
B. 100
0273
OD 0

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 09:40
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1

Chemistry, 23.06.2019 06:00
When hydrogen peroxide (h2o2) is added to potassium iodide (ki) solution, the hydrogen peroxide decomposes into water (h2o) and oxygen (o2). the chemical equation for the decomposition reaction is: 2h2o2—> 2h2o + o2. what is the role of the potassium iodide in this reaction? a. reactant. b. product. c. precipitate. d. catalyst.
Answers: 1

Chemistry, 23.06.2019 06:00
In an exothermic reaction at equilibrium, what is the effect of lowering the temperature? a. the reaction makes more products. b. the reaction makes more reactants. c. the reaction is unchanged.
Answers: 1
You know the right answer?
On the Kelvin temperature scale, which value approximately represents the freezing point of water?...
Questions

English, 30.11.2021 16:30

World Languages, 30.11.2021 16:30


English, 30.11.2021 16:30

English, 30.11.2021 16:30


Business, 30.11.2021 16:30



Mathematics, 30.11.2021 16:30

English, 30.11.2021 16:30

English, 30.11.2021 16:30








Mathematics, 30.11.2021 16:40





