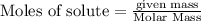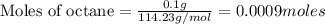Chemistry, 15.12.2020 16:40 BlueExorcistReaper
The enthalpy of combustion of octane is -5470 kJ/mol. Octane (formulae C8H18) was used to heat some water in a copper can. The amount of octane used up was 0.1 g. The amount of water in the can was 100 cm3 . a) Find the moles of octane which was used up. (Show calculations)

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 07:30
In a reaction (at equilibrium) that makes more moles of gas than it consumes, what is the effect of increasing the pressure?
Answers: 1

Chemistry, 22.06.2019 09:00
Scientific evidence tells us that the cause of earths four season is the tilt of earth as it revolves around the sun. the student is instructed to illustrate this information in a science notebook. how will the student illiterate winter in the northern hemisphere?
Answers: 3

Chemistry, 22.06.2019 12:30
Nebulae are enormous clouds in outer space. they are made mostly of hydrogen gas, helium gas, and dust. some nebulae glow brightly, while others do not. the stars that people see are huge, bright balls of glowing gas. they are made mostly of hydrogen and helium. which statement correctly describes other ways in which nebulae and stars are different? a. stars can form inside a nebula but a nebula can never be produced by any star. b. a star always has a higher density than a nebula. c. stars can never form inside a nebula but a nebula can be produced by any star. d. a nebula always has a higher density than a star.
Answers: 3
You know the right answer?
The enthalpy of combustion of octane is -5470 kJ/mol. Octane (formulae C8H18) was used to heat some...
Questions

History, 12.12.2021 23:30


Social Studies, 12.12.2021 23:30

Mathematics, 12.12.2021 23:30

Business, 12.12.2021 23:30

History, 12.12.2021 23:30

Physics, 12.12.2021 23:30



Mathematics, 12.12.2021 23:30

Business, 12.12.2021 23:30



SAT, 12.12.2021 23:30



Mathematics, 12.12.2021 23:30


Mathematics, 12.12.2021 23:30

Mathematics, 12.12.2021 23:30