

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:40
Apiece of copper has a temperature of 75.6 0c. when the metal is placed in 100.0 grams of water at 19.1 0c, the temperature rises by 5.5 0c. what is the mass of the metal?
Answers: 1

Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2

Chemistry, 23.06.2019 04:20
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1

Chemistry, 23.06.2019 12:30
If you reacted 450 g of trimethylgallium with 300 g of arsine, what mass of gaas could you make?
Answers: 1
You know the right answer?
When hydrogen reacts with calcium metal, what are the oxidation numbers of the calcium and hydrogen...
Questions


Arts, 08.02.2021 01:00

Mathematics, 08.02.2021 01:00


Chemistry, 08.02.2021 01:00




Mathematics, 08.02.2021 01:00


Mathematics, 08.02.2021 01:00


Mathematics, 08.02.2021 01:00

Mathematics, 08.02.2021 01:00

Mathematics, 08.02.2021 01:00




History, 08.02.2021 01:00

Mathematics, 08.02.2021 01:00



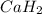 , it is -2.
, it is -2.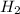 is zero and oxidation number of hydrogen in
is zero and oxidation number of hydrogen in 

