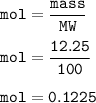Chemistry, 15.12.2020 14:00 royaltyy6533
A scientist completes a thermal decomposition reaction. 12.25g of calcium carbonate, CaCO3, is heated
and forms calcium oxide, Cao, and carbon dioxide, CO2.
a. Write a balanced chemical equation to demonstrate this reaction. Include state symbols.
b. Calculate the number of moles of calcium carbonate that is thermally decomposed in this
reaction.
Ok

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:30
Which change in temperature is the smallest? a change of 1 thomson degree a change of 1 kelvin degree a change of 1 fahrenheit degree a change of 1 celsius degree
Answers: 1

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2

You know the right answer?
A scientist completes a thermal decomposition reaction. 12.25g of calcium carbonate, CaCO3, is heate...
Questions


Mathematics, 08.10.2019 12:30




Biology, 08.10.2019 12:30


English, 08.10.2019 12:30





Biology, 08.10.2019 12:30

Mathematics, 08.10.2019 12:30





History, 08.10.2019 12:30

Social Studies, 08.10.2019 12:30