
Chemistry, 15.12.2020 07:10 arianayoung
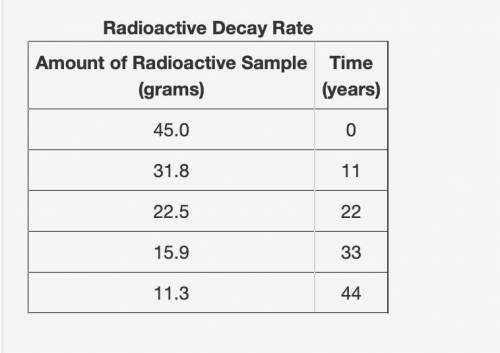
Part 1: What is the half-life of the element? Explain how you determined this.
Part 2: How long would it take 308 g of the sample to decay to 4.8125 grams? Show your work or explain your answer.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:30
You are performing an experiment in a lab to attempt a new method of producing pure elements from compounds. the only problem is that you do not know what element will form. by your previous calculations you know that you will have 6.3 moles of product. when it is complete, you weigh it and determine you have 604.4 grams. what element have you produced?
Answers: 1

Chemistry, 22.06.2019 14:20
You have a liquid that exhibits diltancy. you want to pour it from a bottle. what should you do to the bottle before pouring
Answers: 1

Chemistry, 22.06.2019 17:30
I'm learning about the periodic tables and what each subject's configuration is. for example, hydrogen is 1s^1, but i don't understand how you get that. can someone me understand how to figure out how to figure this out? sorry if the question makes no sense, but it would really a lot if you could me understand! you so much if you can!
Answers: 1

Chemistry, 22.06.2019 23:20
In medium-sized stars such as the sun, nuclear fusion almost always means the fusing of nuclei to form , but larger stars can produce elements as heavy as
Answers: 2
You know the right answer?
Part 1: What is the half-life of the element? Explain how you determined this.
Part 2: How lon...
Part 2: How lon...
Questions



Biology, 07.09.2020 14:01


Physics, 07.09.2020 14:01

English, 07.09.2020 14:01

Mathematics, 07.09.2020 14:01



Mathematics, 07.09.2020 14:01

Computers and Technology, 07.09.2020 14:01

Mathematics, 07.09.2020 14:01


Mathematics, 07.09.2020 14:01



Computers and Technology, 07.09.2020 14:01

Mathematics, 07.09.2020 14:01

Mathematics, 07.09.2020 14:01

Mathematics, 07.09.2020 14:01



