
Chemistry, 15.12.2020 06:10 enrique3300
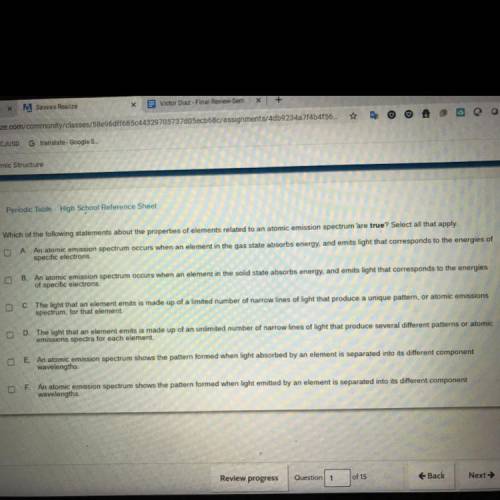
HELP ASAPP!! Which of the following statements about the properties of elements related to an atomic emission spectrum are trueSelect all that apply A An atomic emission spectrum occurs when an element in the gas state absorbs energy, and emits light that corresponds to the energies of specific electrons An atomic emission spectrum occurs when an element in the solid state absorbs energyand emits light that corresponds to the energies of specific electrons The light that an element emits is made up of a limited number of narrow lines of light that produce a unique pattern , atomic emissions spectrum, for that element D The that an element emits is made up an unlimited number of narrow lines light that produce several different patterns or atomic emissions spectra for each element EAn atomic emission spectrum shows the pattern formed when light absorbed by an element is separated into its different component wavelengths An atomic emission spectrum shows the pattern formed when light emitted by an element is separated into its different component wavelengths

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:30
Find the empirical formula of each of the following compounds. given mass or for each element in a sample of the compound 3,611 g ca; 6.389 g c1
Answers: 1

Chemistry, 22.06.2019 02:30
What is the relation between concentration of reactants and the rate of chemical reaction?
Answers: 1


You know the right answer?
HELP ASAPP!! Which of the following statements about the properties of elements related to an atomic...
Questions



















History, 19.11.2019 06:31

History, 19.11.2019 06:31



