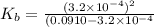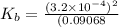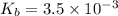Chemistry, 14.12.2020 16:50 bethania26
An unknown weak base with a concentration of 0.0910 M has a pH of 10.50. What is the Kb of this base

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 04:30
Using the periodic table, complete the table to describe each atom. type in your answers
Answers: 3

Chemistry, 22.06.2019 09:00
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1

Chemistry, 22.06.2019 16:50
Which element is least likely to undergo a chemical reaction
Answers: 3
You know the right answer?
An unknown weak base with a concentration of 0.0910 M has a pH of 10.50. What is the Kb of this base...
Questions

Mathematics, 12.12.2021 04:20










English, 12.12.2021 04:20


English, 12.12.2021 04:30

Mathematics, 12.12.2021 04:30

Mathematics, 12.12.2021 04:30




Mathematics, 12.12.2021 04:30

Mathematics, 12.12.2021 04:30

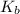 for the weak base is
for the weak base is 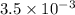
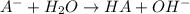
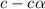
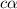
![K_b=\frac{[HA][OH^-]}{[A^-]}](/tpl/images/0980/4654/205fb.png)
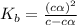
![pOH=-log[OH^-]](/tpl/images/0980/4654/4fd6b.png)
![[OH^-]=antilog(-3.5)= 3.2\times 10^{-4}M](/tpl/images/0980/4654/30b96.png)
![[OH^-]=c\alpha=3.2\times 10^{-4}](/tpl/images/0980/4654/5216a.png)