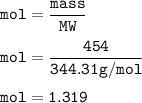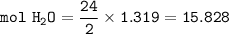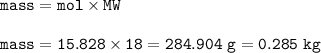Chemistry, 12.12.2020 17:00 miguegen6225
454 grams of isomaltitol, C12H24O11, was combusted in a furnace completely in an excess oxygen. The products of this reaction are water and carbon dioxide. What is the theoretical yield (in kilograms) of water produced from this reaction?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2


Chemistry, 22.06.2019 15:20
Draw any one of the skeletal structures of a 2° alkyl bromide having the molecular formula of c6h13br and two stereogenic centers. indicate chirality by using wedge and hashed wedge notation. lone pairs do not need to be shown.
Answers: 1

Chemistry, 22.06.2019 19:40
What is the wavelength of a 3*10^12 hz infrared wave a 3*10^20m b 1* 10^4m c 3*10^-3m d 1*10^-4 m
Answers: 1
You know the right answer?
454 grams of isomaltitol, C12H24O11, was combusted in a furnace completely in an excess oxygen. The...
Questions


Mathematics, 01.12.2020 20:10

Geography, 01.12.2020 20:10

Social Studies, 01.12.2020 20:10

Social Studies, 01.12.2020 20:10


History, 01.12.2020 20:10





Mathematics, 01.12.2020 20:10

English, 01.12.2020 20:10

Mathematics, 01.12.2020 20:10




English, 01.12.2020 20:10