
Chemistry, 21.09.2019 01:40 dbenjamintheflash5
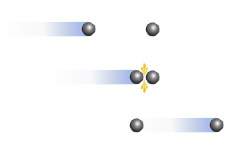
Consider the diagram. which postulate of the kinetic-molecular theory best describes the event in the diagram?
a. most of the volume of a gas is empty space.
b. all collisions between particles are perfectly elastic.
c. there is no force of attraction or repulsion between gas particles.
d. the average kinetic energy of particles depends only on temperature.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
What are the charges of the subatomic particles by choosing the answer from the drop down menu. protons have a (+1,0,or-1). (protons,neutrons,electrons) have a 0 charge. 3.) electrons have a (+1,0,-1)
Answers: 2

Chemistry, 22.06.2019 06:30
Use examples from the article to explain one positive and one negative effect that chemistry has had on society.
Answers: 2

Chemistry, 23.06.2019 06:00
•what conclusions can you make about the relationship between the volume of a gas and its temperature? • what conclusions can you make about the relationship between the volume of a gas and its pressure? • what possible variables have you not accounted for? as you did the procedures, is it possible that the atmospheric pressure may have changed? if it did change over the course of your experiment, then how would your results have been affected?
Answers: 3

Chemistry, 23.06.2019 17:20
What is the relationship between ka and kb with kw? the sum of ka and kb equals the auto-dissociation constant for water. the product of ka and kb equals the auto-dissociation constant for water. the quotient of ka and kb equals the auto-dissociation constant for water. the difference of ka and kb equals the auto-dissociation constant for water.
Answers: 3
You know the right answer?
Consider the diagram. which postulate of the kinetic-molecular theory best describes the event in th...
Questions

Mathematics, 12.11.2020 01:00

English, 12.11.2020 01:00




Computers and Technology, 12.11.2020 01:00

Mathematics, 12.11.2020 01:00


Mathematics, 12.11.2020 01:00



Mathematics, 12.11.2020 01:00



World Languages, 12.11.2020 01:00

Biology, 12.11.2020 01:00



Medicine, 12.11.2020 01:00



