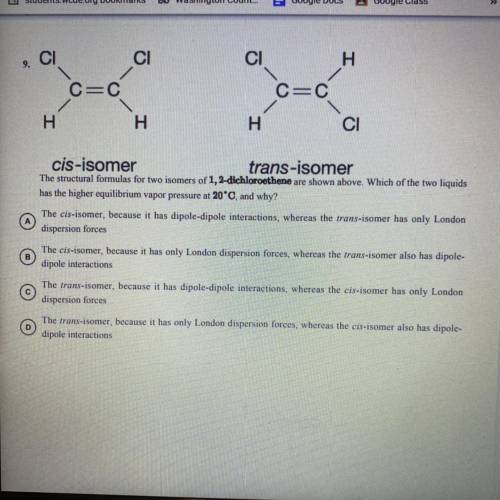
The structural formulas for two isomers of 1, 2-dichloroethene are shown above. Which of the two liquids has the higher equilibrium vapor pressure at 20 degrees Celsius and why?
A) the cis-isomer, because it has dipole-dipole interactions, whereas the trans-isomer has only 1 London dispersion forces.
B) the cis-isomer, because it has only London dispersion forces, whereas the trans-isomer also has dipole-dipole interactions
C) the trans-isomer, because it has dipole-dipole interactions, whereas the cis-isomer has only 1 London dispersion forces
D) the trans-isomer, because it has only 1 London dispersion forces, whereas the cos-isomer also has dipole-dipole interactions

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:30
What’s the scientific notation for the number 6,840,000,000
Answers: 1

Chemistry, 22.06.2019 04:30
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1
You know the right answer?
The structural formulas for two isomers of 1, 2-dichloroethene are shown above. Which of the two liq...
Questions



Biology, 26.07.2020 01:01





Computers and Technology, 26.07.2020 01:01



Computers and Technology, 26.07.2020 01:01







Mathematics, 26.07.2020 01:01


Mathematics, 26.07.2020 01:01