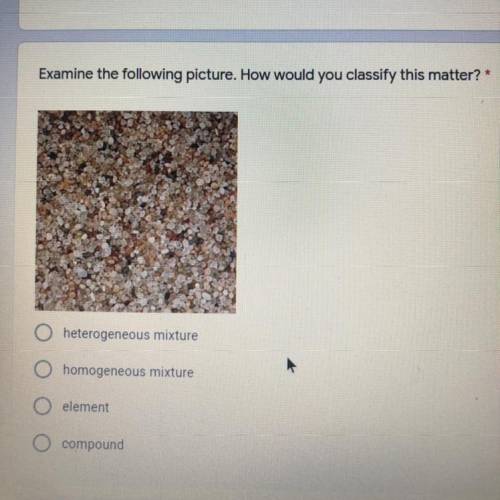
Someone please help me figure this out!!
...

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 13:00
The molality of calcium chloride (cacl2) in an aqueous solution is 2.46 m. what is mole fraction of the solute?
Answers: 3

Chemistry, 22.06.2019 15:00
What is the most important factor in determining climates.
Answers: 1

Chemistry, 23.06.2019 04:40
6) (a) calculate the absorbance of the solution if its concentration is 0.0278 m and its molar extinction coefficient is 35.9 l/(mol cm). the depth of the cell is 5 mm. (b) what is the %t? (7) calculate the absorbance of the solution if the transmitted light intensity is 70% of the initial light beam intensity
Answers: 1
You know the right answer?
Questions

Biology, 19.10.2019 07:50

English, 19.10.2019 07:50




Health, 19.10.2019 07:50

Mathematics, 19.10.2019 07:50



Mathematics, 19.10.2019 07:50


Physics, 19.10.2019 07:50


Biology, 19.10.2019 07:50

English, 19.10.2019 07:50

Mathematics, 19.10.2019 07:50

Mathematics, 19.10.2019 07:50

Geography, 19.10.2019 07:50

Health, 19.10.2019 07:50