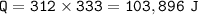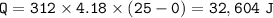Chemistry, 10.12.2020 20:00 marissagirl9893
how many joules are needed to convert 312.0g of ice at -35.0 degrees C to liquid water at 25.0 degrees C.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 2

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 12:30
What is the percent composition of ca(oh)2? 37.7% ca, 53.0% o, and 10.3% h 45.5% ca, 38.2% o, and 16.3% h 54.0% ca, 43.0% o, and 2.7% h 64.7% ca, 27.0% o, and 8.3% h
Answers: 2

Chemistry, 22.06.2019 16:30
An atom with 7 protons, 6 neutrons, and 7 electrons has an atomic mass of amu. (enter a whole number.) numerical answers expected! answer for blank 1:
Answers: 3
You know the right answer?
how many joules are needed to convert 312.0g of ice at -35.0 degrees C to liquid water at 25.0 degre...
Questions

Mathematics, 03.12.2020 19:00

Mathematics, 03.12.2020 19:00



Computers and Technology, 03.12.2020 19:00

Mathematics, 03.12.2020 19:00



Mathematics, 03.12.2020 19:00





English, 03.12.2020 19:00

History, 03.12.2020 19:00


Mathematics, 03.12.2020 19:00

Mathematics, 03.12.2020 19:00

Biology, 03.12.2020 19:00