
Chemistry, 10.12.2020 17:00 antoinewill05
Your Kid Brother's Hint List
1. Group 15 has elements with increasing atomic radii in the order Y, Kc, Jf. C. Nb, and Xt Y is the smallest and Yt is the largest.
2. In the 7th period, the order of decreasing ionization energy is Hn, E. Tu, Xt. Jc. Rc. By, and N. Hn has the largest ionization energy and N has the smallest.
3. Group 14's elements' ionization energies in decreasing order are Sh. Id, Fm. Lb. Me, and Jc Sh has
the largest ionization energy and Jc has the smallest.
4. In the group with 6 valence electrons, the order of increasing electronegativity is Tu, L, M, V. D, and
A. A is the most electronegative, and Tu is the least.
5. The following elements are in the same period but do not include the noble gas. The order of decreasing
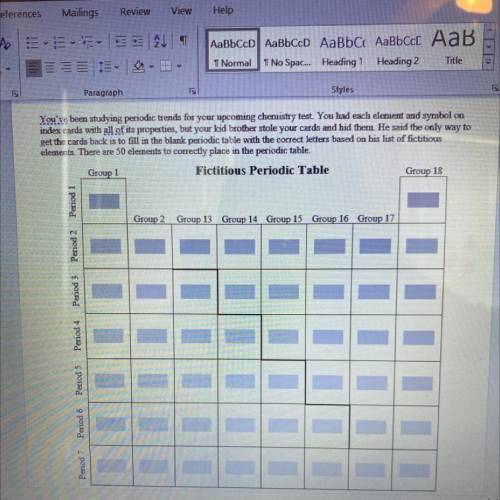

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which type of orbits are found in the principal energy level n = 2 a - s b - s, f c - s, d d - s, p e - s, p, d
Answers: 1


Chemistry, 22.06.2019 19:00
Which change to the system wood cause the freely-moving piston to lower?
Answers: 1

You know the right answer?
Your Kid Brother's Hint List
1. Group 15 has elements with increasing atomic radii in the order Y,...
Questions

Mathematics, 01.10.2021 01:20

Social Studies, 01.10.2021 01:20


Mathematics, 01.10.2021 01:20



Social Studies, 01.10.2021 01:20


Biology, 01.10.2021 01:20

Social Studies, 01.10.2021 01:20

English, 01.10.2021 01:20

Computers and Technology, 01.10.2021 01:20

Mathematics, 01.10.2021 01:20


Mathematics, 01.10.2021 01:20

English, 01.10.2021 01:20

Biology, 01.10.2021 01:20

Mathematics, 01.10.2021 01:20

History, 01.10.2021 01:20




