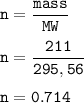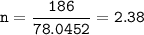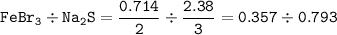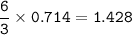A sample of 211 g of iron (III) bromide is reacted with
186 g of sodium sulfide to produce iron (III) sulfide
and sodium bromide. Using the balanced equation
below, predict which is the limiting reactant and the
maximum amount in moles of sodium bromide that
can be produced.
2FeBr3 + 3Na2S → Fe2S3 + 6NaBr

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 13:30
Astudent is trying to create a table that compares hypotheses, theories, and laws. hypothesis theory law do scientific researchers formulate it? yes yes yes does it explain why things happen? yes yes no yes yes yes is it used to make predictions? no yes yes which of the following questions would most likely fill the blank in the table? is it an intelligent guess? is it newly formulated? is it based on observations? has it been proved?
Answers: 1

Chemistry, 23.06.2019 00:30
What is the chemical formula of magnesium bromide? a. mgbr2 b. mgbr c. mg2br2 d. mg2br
Answers: 3

Chemistry, 23.06.2019 03:00
Asample of sea water contains 6.28g of sodium chloride per litre of solution. how many milligrams of sodium chloride would be contained in 15.0ml of this solution?
Answers: 3

Chemistry, 23.06.2019 04:20
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1
You know the right answer?
A sample of 211 g of iron (III) bromide is reacted with
186 g of sodium sulfide to produce iron (II...
Questions


History, 13.07.2019 11:30




History, 13.07.2019 11:30

History, 13.07.2019 11:30


Social Studies, 13.07.2019 11:30


History, 13.07.2019 11:30







Social Studies, 13.07.2019 11:30

English, 13.07.2019 11:30