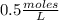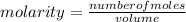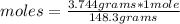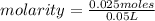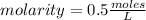Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:30
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10^-3 m and k for the dissociation is 1.86x10^-5. ch3cooh(aq)+h2o(> h3o^+(aq)+ch3coo^-(aq)
Answers: 2

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 07:00
What is the main purpose of patent attorneys? defend the company against legal claims manage financial investments invent new products protect rights to new products and processes
Answers: 1

Chemistry, 22.06.2019 15:50
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate
Answers: 1
You know the right answer?
What is the molar concentration (in moles/L) of a solution made with 3.744 g of Mg(NO3)2 dissolved i...
Questions

Mathematics, 30.06.2019 13:00


Mathematics, 30.06.2019 13:00

History, 30.06.2019 13:00


History, 30.06.2019 13:00


Chemistry, 30.06.2019 13:00



History, 30.06.2019 13:00

Biology, 30.06.2019 13:00

French, 30.06.2019 13:00

Mathematics, 30.06.2019 13:00

Mathematics, 30.06.2019 13:00



Chemistry, 30.06.2019 13:00

Mathematics, 30.06.2019 13:00