Ethane (C2H6) combines with oxygen to
yield carbon dioxide and water.
Let's begin by writing...

Chemistry, 08.12.2020 23:50 McKenzie8409
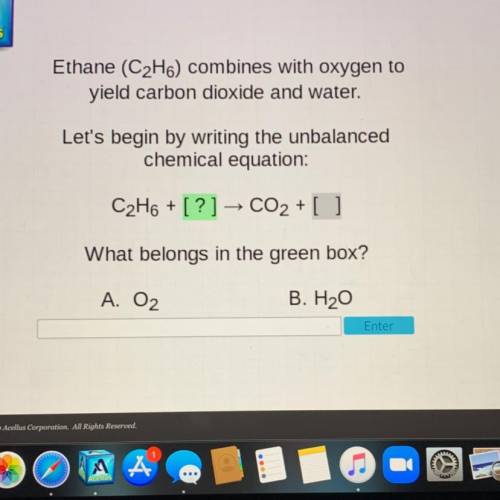
Ethane (C2H6) combines with oxygen to
yield carbon dioxide and water.
Let's begin by writing the unbalanced
chemical equation:

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Write the empirical chemical formula of calcium with a mass percent of 38.8, phosphorus with a mass percent of 20.0, and oxygen with a mass percent of 41.3.
Answers: 1

Chemistry, 22.06.2019 11:00
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1

Chemistry, 22.06.2019 12:00
What does a complete balanced chemical equation include? a. exothermic coefficients b. endothermic coefficients c. valence electrons d. molar coefficients
Answers: 1

Chemistry, 22.06.2019 13:00
The number of neutrons is equal to the atomic number minus the atomic mass. a. true b. false
Answers: 2
You know the right answer?
Questions


Mathematics, 21.11.2020 21:50

Chemistry, 21.11.2020 21:50







Biology, 21.11.2020 21:50

Mathematics, 21.11.2020 21:50

English, 21.11.2020 21:50

Social Studies, 21.11.2020 21:50

Mathematics, 21.11.2020 21:50


Arts, 21.11.2020 21:50


Mathematics, 21.11.2020 21:50

Mathematics, 21.11.2020 21:50

Biology, 21.11.2020 21:50



