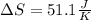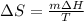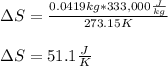Chemistry, 08.12.2020 14:00 choiboiqg5755
Calculate the change in entropy as 0.0419 kg of ice at 273.15 K melts. The latent heat of fusion of water is 333000 J/kg .

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Consider the nuclear equation below. 239 > x + 4 he 94 2 what is x? 1.235 cm 96 2.243 u 92 3.235 u 92 4.243 cm 96
Answers: 2

Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1

Chemistry, 22.06.2019 16:40
Identify the lewis acid in this balanced equation: ag+ + 2nh3 -> ag(nh3)2+a. ag+b. nh3c. ag(nh3)2+
Answers: 1

You know the right answer?
Calculate the change in entropy as 0.0419 kg of ice at 273.15 K melts. The latent heat of fusion of...
Questions


Advanced Placement (AP), 14.12.2020 22:30




Mathematics, 14.12.2020 22:30


History, 14.12.2020 22:30



History, 14.12.2020 22:30

Chemistry, 14.12.2020 22:30




Mathematics, 14.12.2020 22:30

Mathematics, 14.12.2020 22:30


Mathematics, 14.12.2020 22:30